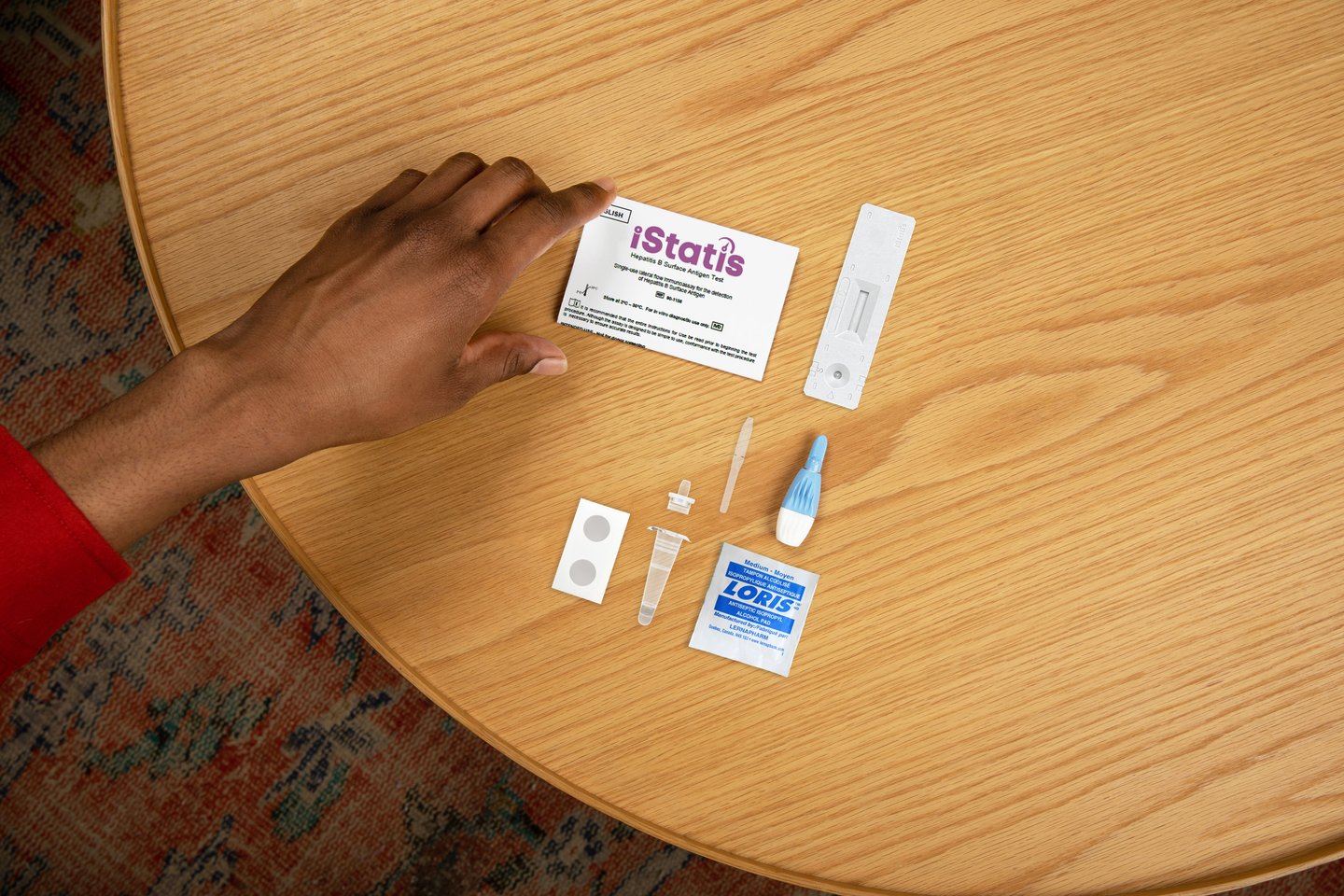
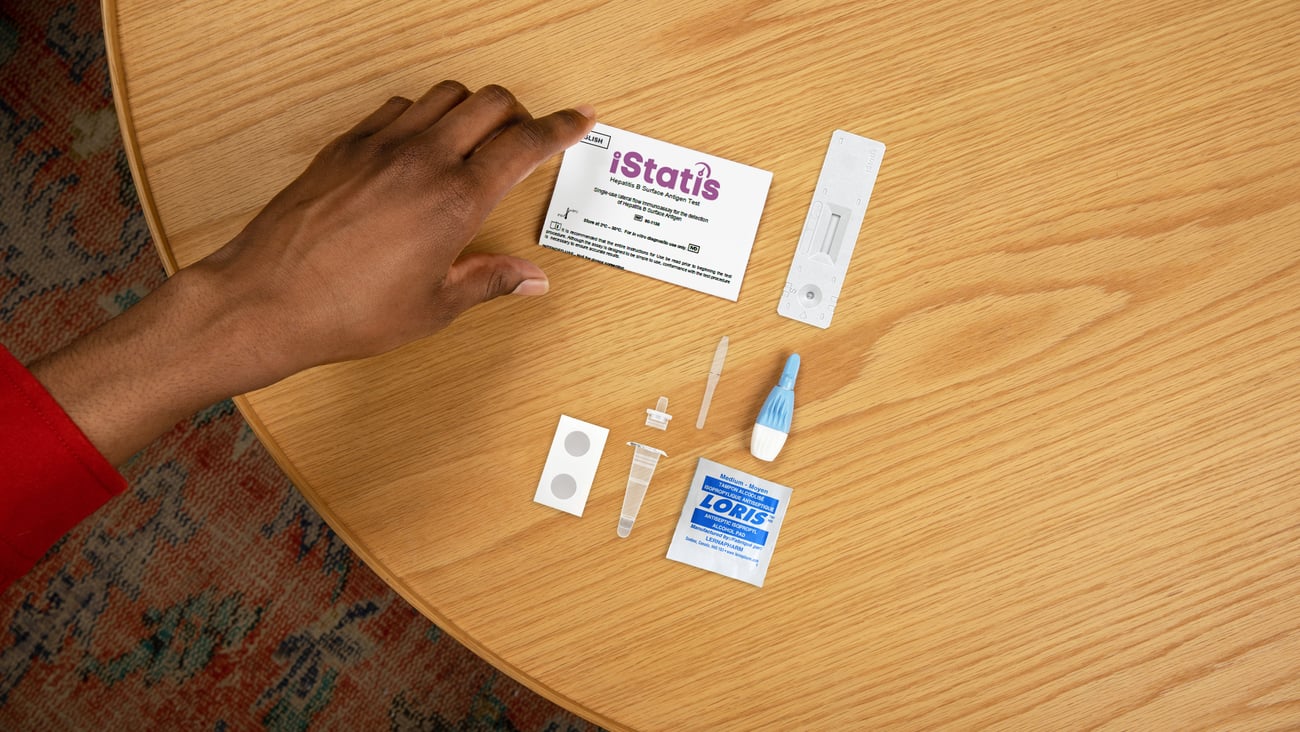
NEW PRODUCT: bioLytical’s iStatis Hepatitis B Surface Antigen Test
British Columbia-based biotechnology company bioLytical Laboratories has announced its hepatitis B test has received Health Canada approval—the first of its kind on the continent.
The iStatis Hepatitis B Surface Antigen Test is North America’s first rapid point-of-care test for hepatitis B. The tests work by detecting hepatitis B surface antigen from a fingerstick blood sample and can deliver results within 15 minutes.
The company notes that although more than 260,000 Canadians are living with chronic hepatitis B, nearly half are unaware of the infection status, and that access to testing continues to be limited in particularly in remote, underserved, and high-risk communities. bioLytical Laboratories say the ease of use of this test makes it ideal for clinics, mobile units and community care settings where traditional lab testing may be inaccessible.
“Too many people with hepatitis B remain undiagnosed because testing isn’t always accessible,” said Robert Mackie, CEO of bioLytical Laboratories, in a news release. “This authorization allows screening to move closer to the patient, whether that’s in a clinic, a mobile unit, or a community outreach setting.”